The Cytel COVID-19 Trial Tracker brings you an up to the minute, real time dashboard about COVID-19 trials around the world. This snapshot gives you a quick briefing on the current state of COVID-19 therapy and vaccines development.
As we head towards the end of 2020, there are 2516 registered trials as on December 8, 2020.
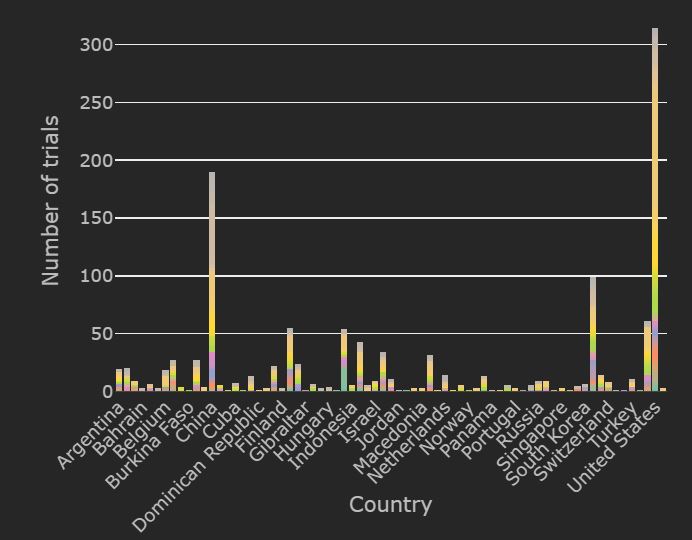
Currently, about 1278 trials are recruiting globally and 309 have completed. Most of the trials currently recruiting are based in the United States and Spain. The top therapies recruiting in the United States are Plasma based therapy, mAb and hydroxychloroquine.
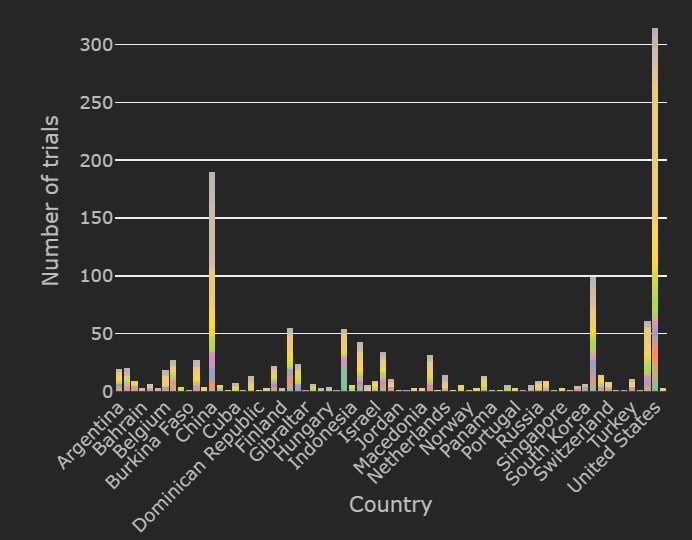
Spain is now leading the pack in numbers of trials for hydroxychloroquine and Tocilizumab trials. Most of the recruiting for Plasma based therapy and Vaccine trials is being done in the United States. Also, approximately 26% of all trials globally that are recruiting currently are based in the United States.
For more information on the Cytel COVID-19 Trial Tracker click below.

Cytel and Ingress Health (now a Cytel company) conducted a study aimed to describe the landscape of the registered COVID-19 clinical trials in terms of trial designs, interventions and endpoints. The core of the analysis was a mapping of the ongoing curative and preventive intervention trials on COVID-19 against the guidelines provided by FDA. Data on trial characteristics were extracted from Cytel Global Coronavirus COVID-19 Clinical Trial Tracker and complemented by other databases (e.g. clinicaltrial.gov). Read our previous blog here to learn more.
Cytel is helping our clients by providing recommendations for completing data collection, handling missing information, modifying protocols and amending statistical analysis plans. In addition, Cytel’s software is helping customers more efficiently design adaptive trials for COVID-19, more accurately analyze small or skewed datasets, and forecast future enrollment for all ongoing trials in the face of so much uncertainty.
Click on the button to learn about our solutions.