Cytel's blog featuring the latest industry insights.

At the close of May 2020, we have about 500 new trials globally but trends in trial design and choice of therapies tested have largely remained the same. The state of Vaccine Trials though has changed in both number and trial design.

At 1265 clinical trials registered around the world, May 2020 has seen approximately 500 new trials registered. commence. Approximately 20% of these trials are Hydroxychloroquine trials (255 out of 1265). The second most popular trials are plasma based therapies, which account for a little over 7% of all trials (92 out of 1265).
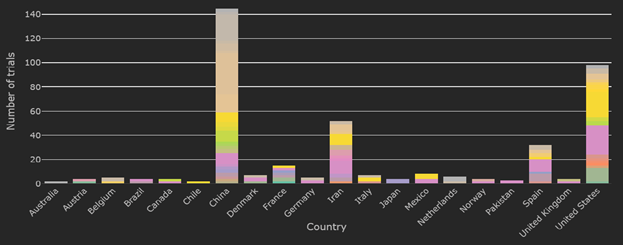
We also continue to see a disparity between registered trials and trials that are recruiting. At present the 732 trials recruiting account for about 57% of all registered trials. About 20% of those recruiting are also Hydroxychloroquine trials (151 out of 732).
Clearly Hydroxychloroquine trials continue to dominate the COVID-19 therapy space. Unsurprisingly, randomized 2-arm trials also dominate COVID-19 therapy development with nearly 600 of the 732 trials currently recruiting being randomized trials and about 500 having 2-arm designs.
At the close of May 2020, China, Iran and the United States continue to have the highest number of trials currently recruiting, with Spain and France leading trial recruitment in the European Union.
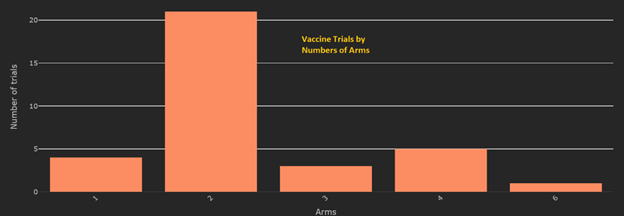
May 2020 though has seen also witnessed an incredible increase in numbers of vaccine trials and a variety in their design. While there were only about 15 registered vaccine trials at the beginning of the month, there are now more than double that number (36 in total). While the majority are 2-arm trials, vaccine trials with their complex innovative designs now include several 3-arm and 4-arm trials, some single arm trials and even a 6-arm trial. They are now recruiting across 13 countries.
Learn more:
