Reinventing Clinical Trial Design: Digital Development

New medicines and devices under development live and die on the strength of their clinical data. An asset’s journey is long and difficult—from assessing tolerability in a first in human study, to proof of pharmacodynamic activity, to proof of clinical activity, to pivotal confirmatory trials for safety and efficacy. At every stage, the data readout from a clinical trial is highly visible for teams, companies, and their investors, and can make or break the project. In this context, planning the right trial is an immensely important endeavor, and integrating strategy and efficient statistical tactics (i.e., intelligent design) even more so. Are your designs and studies the best they can be?
Clinical development professionals are faced with many questions to answer and problems to solve during the product development expedition. What clinical trial design will provide the right answers at the right time? How can it be accomplished quickly and economically?
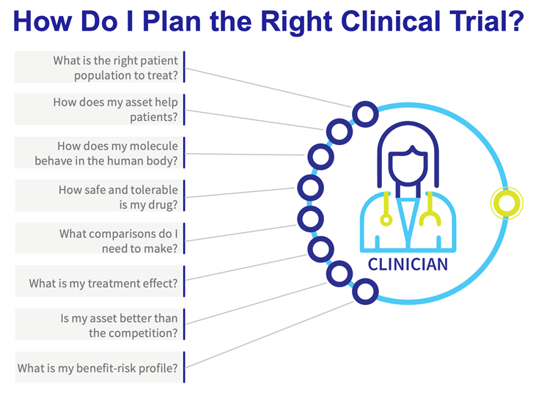
Clinical development professionals are faced with many questions from different stakeholders throughout product development
One of the greatest hindrances in today’s clinical trial design status quo is the tedious manual process. A few very smart people will gather to outline some ideas using antiquated tools like word processors and slideshow applications. Using mostly words on a page, the team will cycle through ideas slowly between roles like clinician and statistician and examine a few designs with different characteristics over several weeks. This iterative sequence typically involves many emails, documents, custom statistical coding modules, and discussions around sample size, power, target values, and their statistical operating characteristics. Invariably there are multiple perspectives on any team and occasionally conflicting agendas. I know from personal experience that misunderstandings are common and that it’s difficult to understand tradeoffs and align everyone’s goals.
There is a better way. Here at Cytel, we’ve created Solara® – a first-in-class digital platform for clinical trial design, strategy, and development. Solara combines 3 things:
- Your team’s inputs
- Cytel’s time tested statistical algorithms (the same as in our East software used by dozens of industry partners and regulators), and
- Massive cloud compute power
Like a high throughput screen for your clinical trial, Solara churns through hundreds of thousands of designs and their associated Monte Carlo simulations in minutes.
This platform empowers clinical development teams to explore, prioritize, and decide together during clinical trial design. Instead of struggling to understand why powering for a given endpoint might not result in the observed target value you want, you and your team can focus on more intuitive design. Specific visualizations show power, sample size, and cost projections in plain language. Tradeoffs between power, cost, and speed are tangible and easily understandable. Implications for study timelines are explicitly graphed. Most powerfully, your team can envision specific risk scenarios, e.g., slow enrollment and unexpected treatment effect (too low or high), via simulation and see how these scenarios affect your clinical trial’s performance. These capabilities meaningfully enhance a team’s design intelligence.
Clinical development is fundamentally a team sport, so while initial versions of Solara were created by statisticians for statisticians (see more details here), Solara is now evolving to fit a clinician’s mindset to help involve the rest of the team. Specifically for clinicians, we have designed a feature set that caters to their workflow and helps address their specific challenges. This all comes together in an efficient and intuitive user interface that helps a clinician focus on applying the art of design and strategy rather than the technique.
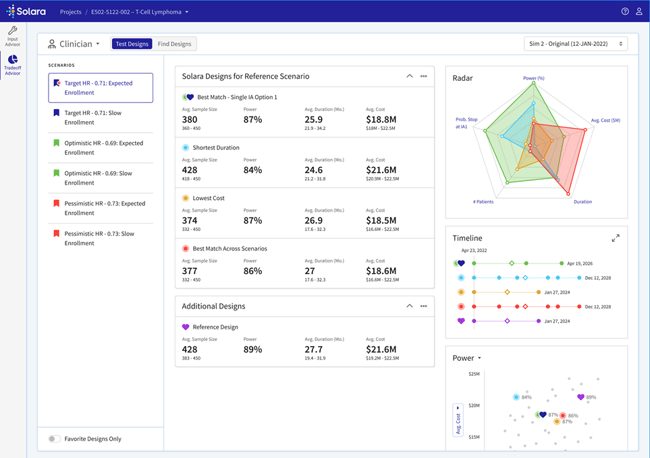
Solara’s Clinician Dashboard is an efficient and intuitive interface to examine trial design options from a clinical perspective
To plan the right trial, the manual process that teams conventionally use is inadequate. Fortunately, there are better tools available in today’s digital age that reimagine clinical trial design as a more nimble endeavor. By letting you focus on strategy and success rather than technical distractions, the Solara platform can help your team see all pertinent paths for your clinical trial’s journey and quickly help find the right one for your needs. Intelligent design, by design.
Live Webinar: Reinventing Trial Design in the Digital Era
Interested in learning more about the new digital approach to clinical development from Albert? Join Cytel's upcoming webinar!
About Albert Kim
 Albert Kim, MD, PhD, is the Chief Medical Officer at Cytel. Dr Kim has rich expertise in cardiovascular disease, translational medicine, and drug development. After receiving degrees from Harvard and UCLA, completing residency at Brigham and Women's Hospital, and undertaking further specialty training at Mass General Hospital and UCSF, he achieved board certification in internal medicine, cardiology, and clinical cardiac electrophysiology.
Albert Kim, MD, PhD, is the Chief Medical Officer at Cytel. Dr Kim has rich expertise in cardiovascular disease, translational medicine, and drug development. After receiving degrees from Harvard and UCLA, completing residency at Brigham and Women's Hospital, and undertaking further specialty training at Mass General Hospital and UCSF, he achieved board certification in internal medicine, cardiology, and clinical cardiac electrophysiology.
Most recently, he was Vice President, Clinical Research Head for Pfizer’s Internal Medicine Research Unit. His roles at Pfizer and at Novartis saw him lead early and late phase programs for common and rare diseases. Dr Kim has contributed to development efforts for several candidate medicines utilizing different therapeutic modalities and has authored a body of work including numerous publications in leading medical journals. He was also a medical reviewer in the Division of Cardiac Devices at the FDA.


