Cytel's blog featuring the latest industry insights.

At a recent PhUSE SDE, Cytel’s Chitra Tirodkar presented how East PROC MCPMod could be used to help solve the problem of uncertain true dose-response relationship in a bronchodilator study. In this blog we summarize some of the issues, and make Chitra's slides available for download.
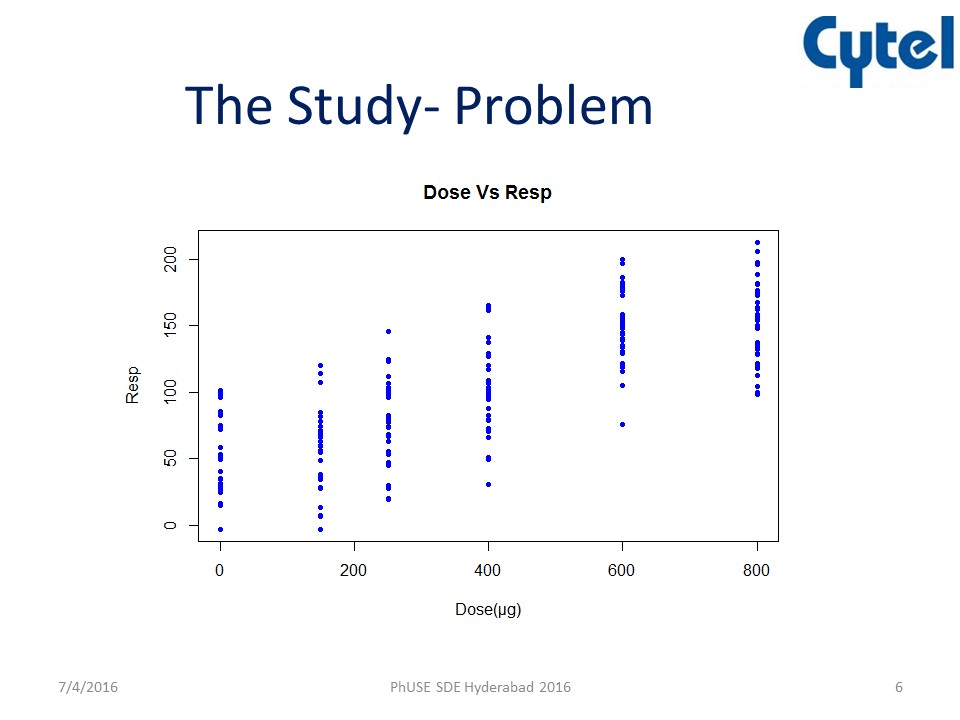
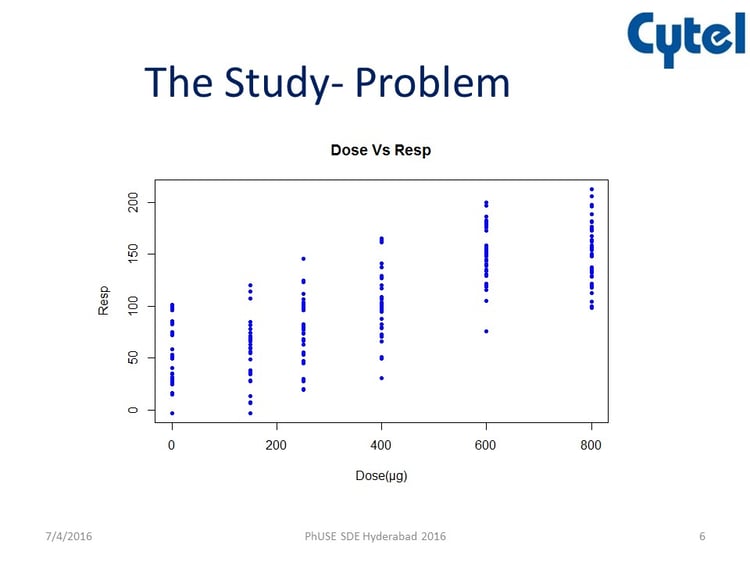
In the presentation, Chitra examined the example of a clinical trial in the area of asthma. The aims of the study were to estimate the bronchodilator dose-effect in pre-schoolers and determine the minimum dose to be used in a bronchodilator test to demonstrate bronchial reversibility. The study background is outlined below.

The problem was that there are no data on Bronchodilator dose-response relationship in wheezy pre-school children whose disease pathology is poorly understood. Indeed, there is a great deal of uncertainty about the dose response relationship.
A traditional approach to such an early phase trial would tackle dose-selection as a series of three steps: proof-of-concept to establish dose-signal (i.e. the existence of a dose-response); dose-response modeling to determine the shape of an entire dose-response curve (e.g. for making predictions); and dose-selection to determine which candidate doses should move forward.
The MCP-Mod approach by contrast, combines multiple comparison and model based approaches and overcomes some of the limitations of traditional approaches in that it is robust to model misspecification and allows for flexible dose estimation. The approach has been endorsed by the FDA and EMA and where appropriate, its use can lead to more informative Phase 2 designs.
In this slide deck presentation, Chitra shares PROC MCPMod outputs generated in relation to this project.
To download the slide presentation click below:
The East PROC MCPMod is the only SAS tool for MCPMOD analysis and can be used seamlessly with other/ existing SAS procedures. To find out more about this tool click below:
Other Cytel Blogs on this Topic
MCPMod for the Modern Dose Ranging Trial
With thanks to Chitra Tirodkar
