Phase 2 Designs for Clinical Utility Limiting Therapies
When testing certain types of new drugs it is known in advance that the adverse side-effects of the medication will limit dose selection. For example, it is well-established that for many new pain medications, the side effects of nausea and vomiting will place constraints on the selection of higher dose levels.
Such therapies are sometimes called clinical utility limiting therapies, where clinical utility refers to a benefit-risk assessment typically calculated using a function of efficacy and safety parameters. When tolerability decreases rapidly with higher level doses, obtaining high clinical utility is only possible for a small spectrum of doses. Where this spectrum falls is difficult to determine without a dose-finding clinical trial.
Such a scenario for dose-finding creates a special problem for the well-being of patients participating in said clinical trials: When using a conventional trial design for pain medication, most patients will be randomized into trial arms where the dose will either be too low to relieve pain, or too high for tolerability. The fact that many patients could be randomized into such arms, calls for investigation into improved trial designs where the majority of patients are randomized into treatment arms in the vicinity of the dose with optimal clinical utility. The challenge, of course, is that the trial design has to be constructed without any knowledge of what the right dose actually is.
Enter the Adaptive Maximizing Design for Clinical Utility Limiting Therapies.
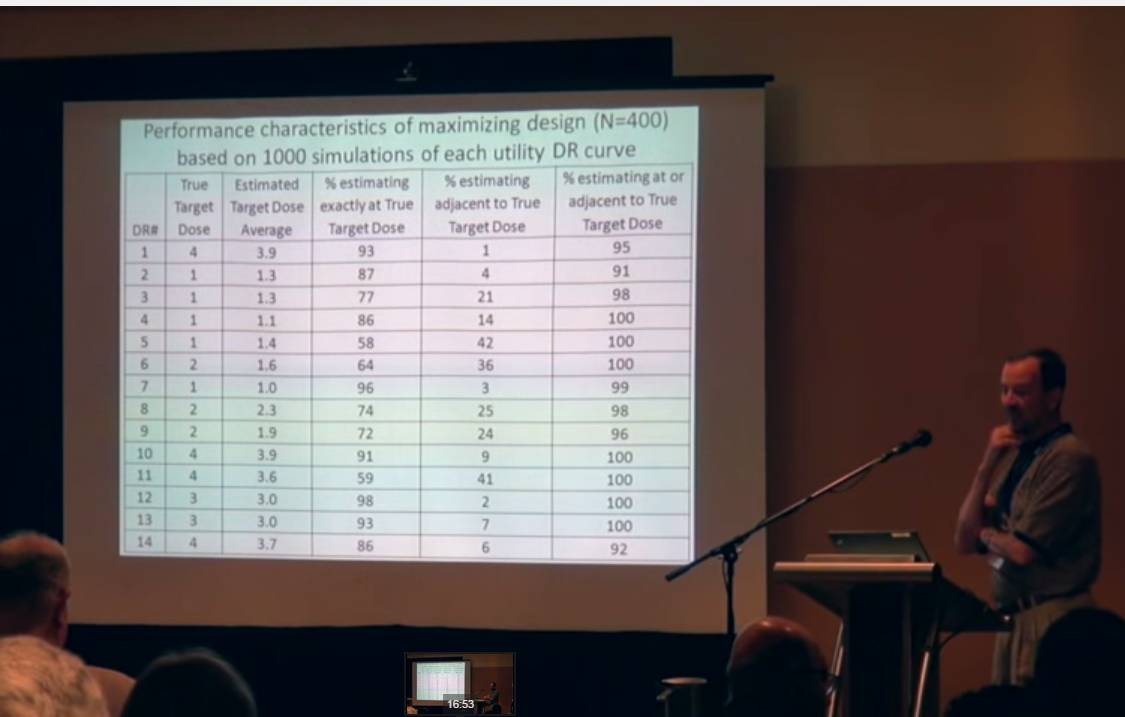
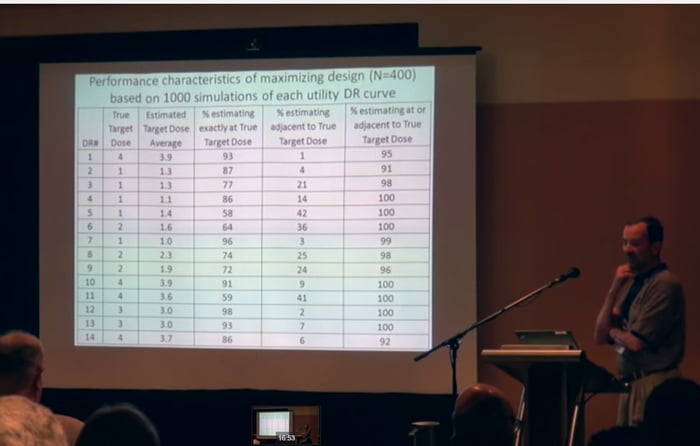
According to a new paper in Therapeutic Innovation & Regulatory Science, co-authored by Cytel Consultant Jim Bolognese, the Adaptive Maximizing Design [AMD] is a modern clinical trial design developed for this specific purpose. Statistically it is constructed to “maximize assignment of patients to doses that demonstrate maximum balance between efficacy and tolerability (i.e., CU) assessed via a CU function.” Jim’s new paper, tests whether this is really the case, using simulations to design a Phase 2 study of Trevena's investigational drug TRV-130 for post-operative pain.
According to the paper’s results, the AMD revealed a “strong capacity” to determine which dose-regimens come closest to maximizing clinical utility with 91% of simulations choosing the right dose of TRV-130. Needless to say that such a design must also have strategic benefits for trial sponsors. The capacity to estimate clinical utility maximizing assignment of patients at or near doses with optimal clinical utility, clearly increases the chances of selecting the correct dose for Phase 3.
Related Items of Interest
Evaluation of an Adaptive Maximizing Design Study Based on Clinical Utility Versus Morphine for TRV130 Proof-of-Concept and Dose-Regimen Finding in Patients
Paper Abstract:
Conventional opioids provide powerful analgesia but also produce efficacy-limiting adverse effects, limiting their clinical utility (CU). TRV130 is being evaluated to determine if CU can be expanded by way of increased efficacy, decreased adverse effects, or some combination thereof. This phase 2 study of TRV130 blends traditional objectives with novel design features aimed toward the specific strategic goal to optimize the attributes of TRV130 efficacy and tolerability compared to the conventional opioid, morphine. The adaptive maximizing design (AMD) was developed to maximize assignment of future patients to doses that demonstrate maximum balance between efficacy and tolerability (i.e., CU) assessed via a CU function. Our evaluation of the AMD performance characteristics reveals that the AMD has a strong capacity to estimate the TRV130 dose-regimens with maximum CU, assign more patients to the TRV130 dose-regimens with maximum CU, and, conversely, assign fewer patients to doses away from those with maximum or near-maximum CU.
Jim discusses his findings below:

