Cytel's blog featuring the latest industry insights.

.png?width=405&height=228&name=Variable_Clusters_(Elashoff_Blog).png)
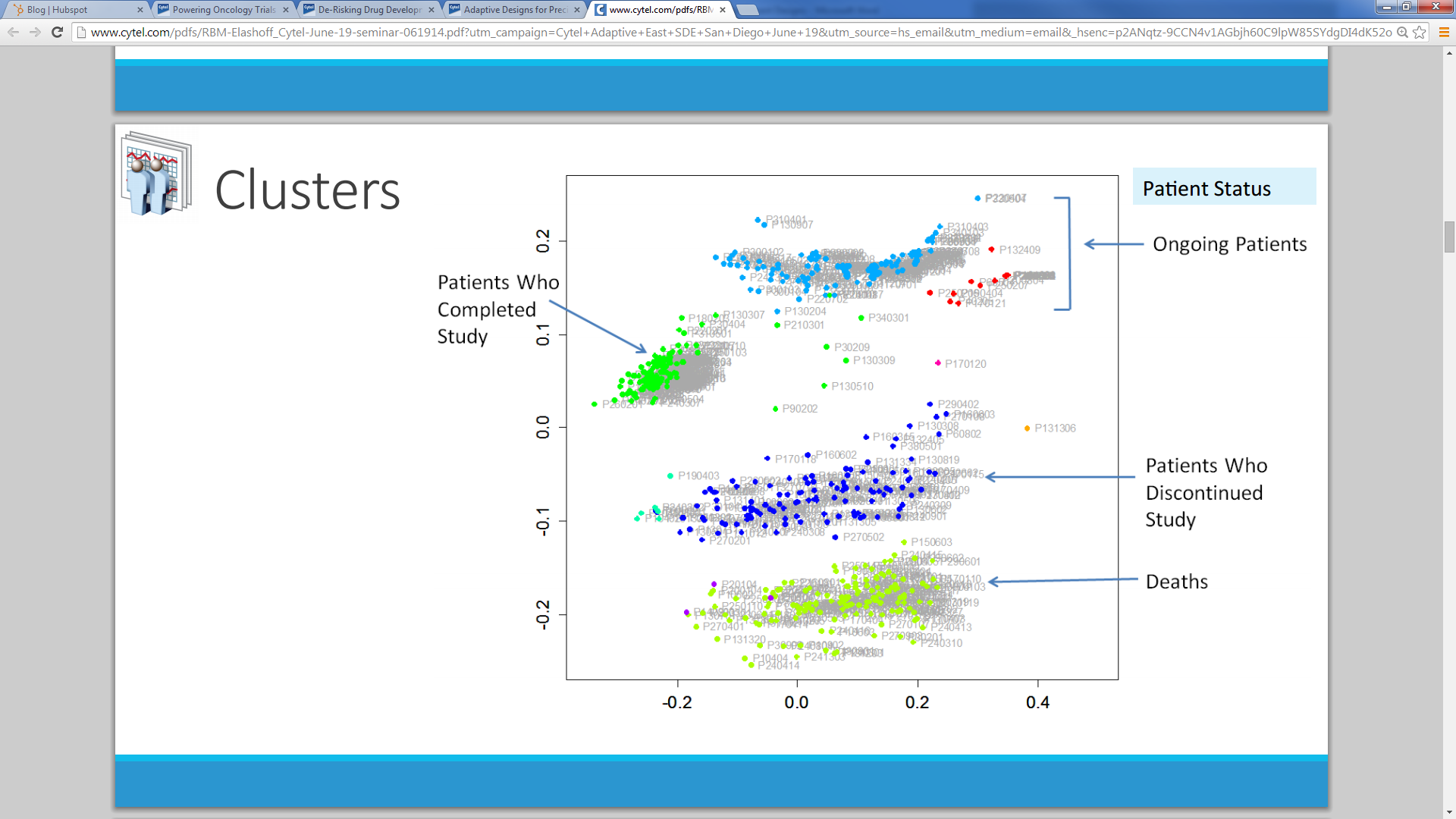
A recent Cytel Seminar on Adaptive Statistical Designs featured a talk by Michael Elashoff (Patient Profiles) on Multivariate Approaches for Risk-Based Monitoring. Elashoff, a former statistical reviewer at the Food and Drug Administration, recommended combining cluster and rules based methods for statistical monitoring. Such adaptive monitoring approaches can substantially reduce the time and expense of data monitoring while ensuring consistently high data quality.
Elashoff recommends using multi-dimensional scaling techniques to co-cluster patients and variables based on the full set of clinical variables collected in a trial. Then for each pair of variables in a cluster, regression models can be used to determine rules that locate outliers in the study data sets. More than 2000 such rules can exist for any given study.
According to Elashoff, the primary benefit of this approach is that it allows clinical data monitors to adapt monitoring techniques as data arrive over the course of a trial. For example, sites that have higher than average outlier rates can have monitoring visits scheduled, and case report forms with excess outliers can undergo scrutiny and revision. Such a procedure maintains high data quality while preserving critical resources in time and expense.
*Several thanks to Michael Elashoff for providing the seminar, the slides and comments on this blog post.
Related Items of Interest
Cytel Seminar on Adaptive Designs for Precision Medicine (Pfizer's Xalkori Trial)
De-Risking Drug Development Using Adaptive Designs (VALOR Trial, Adaptive Sample Size Re-estimation)
Powering Oncology Trials for Success: Adaptive Designs in East (by Cytel Statistician Charles Liu)
