An Interview with Louis Dron on the Benefits and Future of Cytel’s Trial Tracker
The COVID-19 Pandemic prompted the rapid surge in the generation of clinical data that has been scattered across multiple platforms, making it challenging to measure comparative treatment effects across trials. Last year, Cytel launched a COVID-19 Trial Tracker, an Open Access tool to track the global response to the pandemic. We talk to Louis Dron, Director - Real World Analytics at Cytel, about the evolution of Cytel’s Trial Tracker and the vision for its future developments.
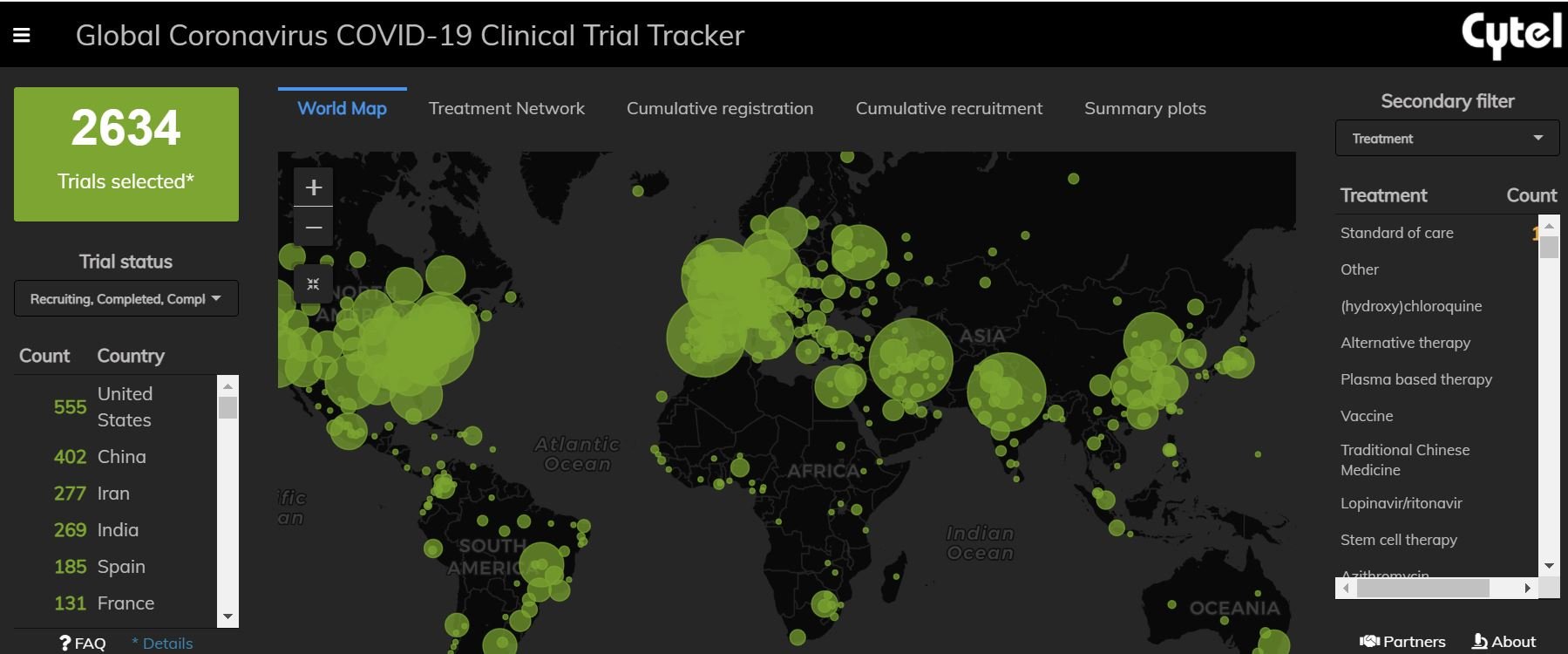
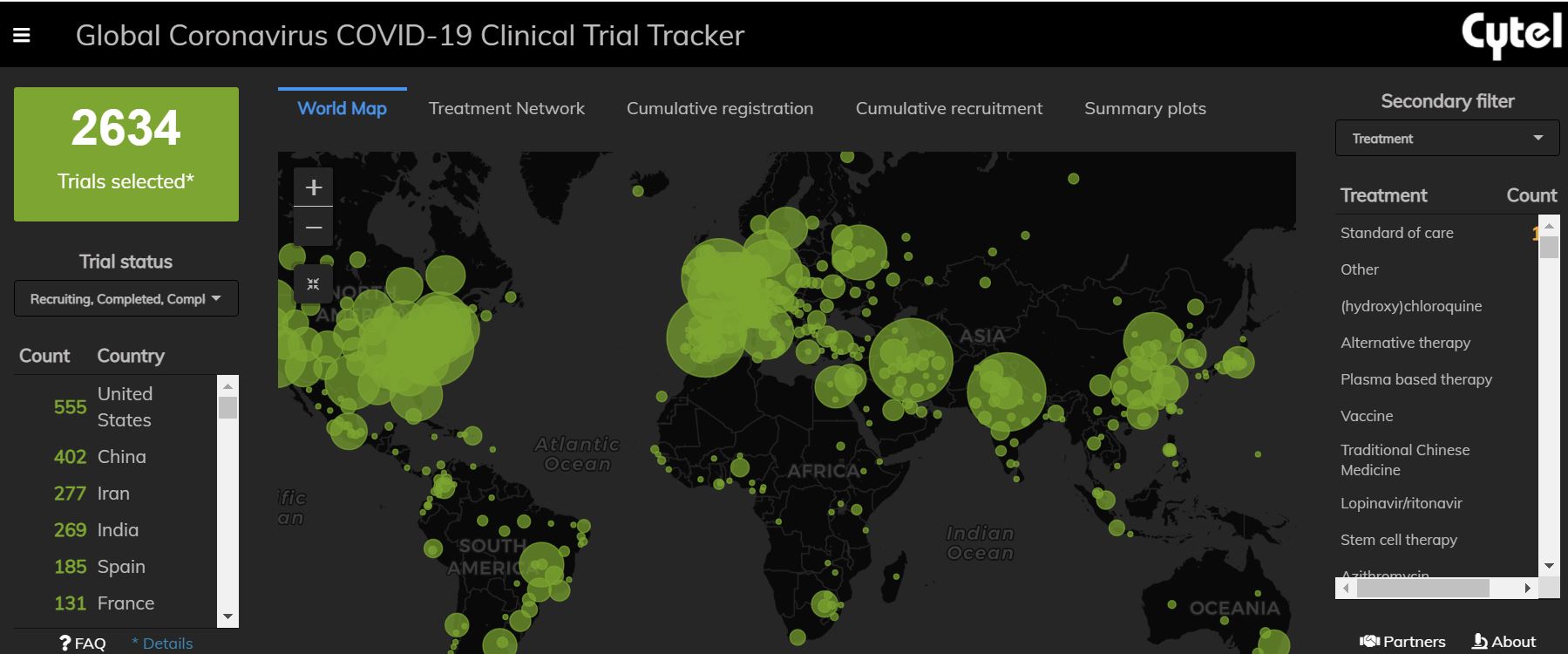
Cytel successfully launched the COVID-19 Clinical Trial Tracker in April 2020. How did we achieve this in such a short span of time?
After taking stock of the situation, we quickly divided work into separate streams to help A) identify data sources, B) understand how to synthesize and interpret information, and C) analyze and visualize the available data. Aspects of our work that we had undertaken prior to this project greatly helped us in this process. Our team has worked extensively in evidence synthesis and many of the approaches we have utilized for a fast start-up were inspired by systematic review processes. In tandem, we have worked extensively in automation of these methods which allowed us to develop near-term and long-term processes for managing the volume of data that was generated. The teams at Cytel collaborate very well with each other and encourage exchange of feedback. Moreover, our contacts in the global research networks allowed us to better understand the needs of the prospective users before the launch of the Trial Tracker.
How has the project evolved over the past year?
Our project was initially intended to provide quick connections for researchers to help minimize duplication of efforts and to better coordinate research activities. Over the past year, it has evolved into a timeline of research conducted in times of the COVID-19 pandemic. With the help of Cytel’s Trial Tracker, we can observe the rise (and fall) of hydroxychloroquine-based trials, the rollout of vaccination programs and the development of targeted novel therapies.
Who are the primary users of the Trial Tracker and what advantages are they seeing in using the tool?
The primary users are a mixture of professional groups looking at understanding what research currently exists and identifying the gaps. Examples include not-for-profit global organizations as well as individuals who have reached out to our team to query certain data trends. Our Trial Tracker has been funded in part by The Bill and Melinda Gates Foundation, a leader in global health solutions. We have collaborated closely with the Gates Foundation throughout the project and more recently with the International COVID Data Alliance (ICODA).
Our contact form has also been a great way to interact with the research community and we have been able to collaborate on research papers as well as provide media outlets with data for their reporting.
Can the framework of this application be used in other contexts?
Very much so! There are many themes to COVID research which are not limited to this disease alone. An overlap of patient populations, recruitment challenges and geographical gaps in evidence are common themes among many well-studied disease topics. We have engaged in early-stage prototype discussions with several groups to explore how evidence mapping in this manner may help align global strategies and identify research gaps.
Do we have any upcoming changes/updates planned for the Trial Tracker?
Aside from routine updates (many trials continue to be registered to this day!), we are exploring how to better identify trials for emerging COVID-research topics such as “long-COVID” and how to integrate our data into other worldwide data synthesis efforts. The ongoing pandemic has provided an opportunity for many fantastic collaborations and our researchers are working with many groups to continue to improve research focus. We are, of course, always eager to hear feedback or field questions from anyone involved in this space and would encourage users to continue to submit their queries via the associated web-page!
The Clinical Trial Tracker has a great visual showcasing a treatment network for a given therapy. Could you quickly explain what the importance of knowing a treatment network is to a non-scientist?
When we think about treatments for a disease, we often want to know which treatment is "best" for a given outcome. Often, trials are compared to either a placebo-controlled group or a group who receives the current "standard-of-care". Other times, trials may compare two or more active interventions to one another. If we want to see how two treatments compare, we can ideally review the results of a head-to-head trial. The visualization of a treatment network can show you what treatments have been directly compared, and how often this has been done. This allows researchers to review where there may be overlap (such as, where there are already over 10 trials investigating a particular head-to-head comparison). Alternatively, there are methods of analysis known as indirect treatment comparison, which allow researchers to estimate how treatments compare when they have not been directly tested in a head-to-head trial. This typically necessitates treatments having a shared comparator, and the treatment network can help identify this.
Could you talk a little bit about the country-level plots. What was this meant to highlight and were there any surprises in looking at the data this way?
Country-level plots provide a unique insight into which countries are focusing on specific treatment classes and where the distribution of this research is occurring. Research even at a within-country level can often be imbalanced with regards to where the research occurs. Throughout the COVID-19 pandemic, we have observed many instances of significant within-country variation in disease burden and research activity. In part, the country plots can provide researchers with a more granular look at their local research efforts to identify gaps or areas where they may contribute. For larger international organizations, country plots present a mechanism to compare a country’s effort in terms of their relative research efforts and allow for resource planning accordingly.
Click the button to access Cytel's Trial Tracker.


