Cytel's blog featuring the latest industry insights.

Most people familiar with adaptive clinical trial designs are familiar with those statistical designs that reject the null hypothesis. These include now familiar designs like the promising zone design and the adaptive switch design.
A newer breed of adaptive designs, however, aims to apply adaptation techniques to confidence intervals.
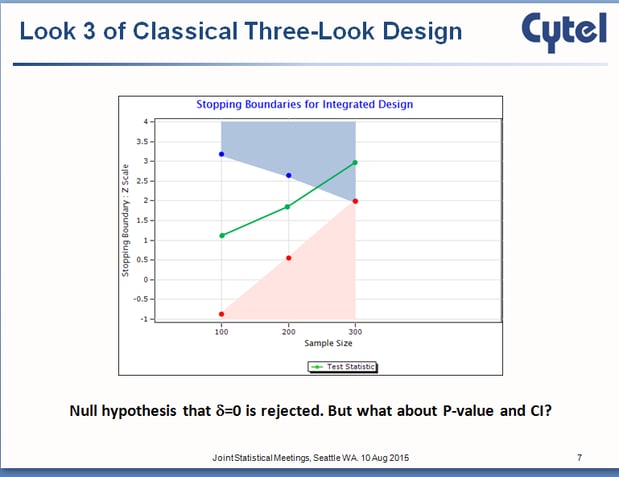
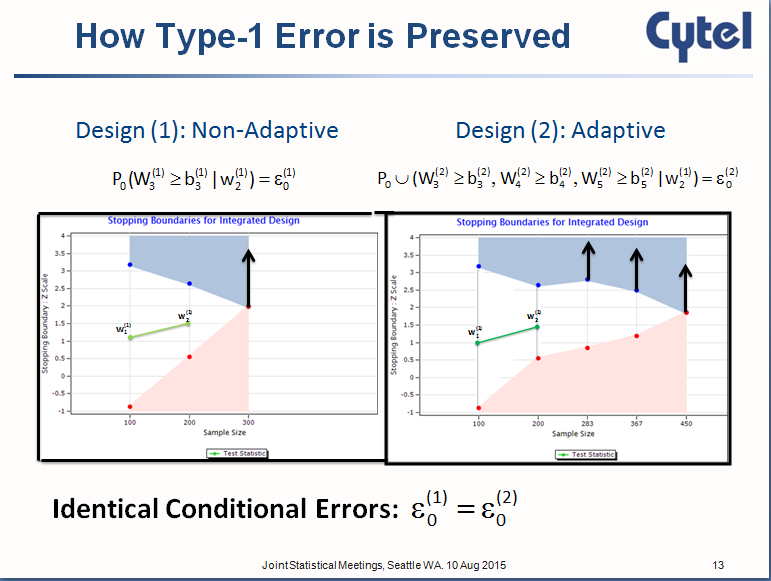
Imagine, as in the graphic, that on a third interim look, it becomes clear that the null hypothesis has been rejected. In a non-adaptive clinical trial design, there is nothing left to do at this point.
Compare this graphicwith the one underneath this paragraph. The design to the left has the original stopping boundaries while the design to the right is a clinical trial design with an adaptive stopping boundary. The adaptation of the boundary takes place after the null hypothesis is rejected.

In 2013 Statistics and Medicine published an article on how to extend the method for stage wise confidence interval or SWCI to designs for adaptive clinical trials [1]. The method, initially developed by Brannath, Mehta and Posch in 2009, was the first to provide exact coverage within an adaptive group sequential setting [2]. The 2013 article extends the method to handle two sided confidence interval tests.
This new method, called backward image confidence intervals or BWCI, is the only published method with exact two-sided coverage.
In the attached slides, Cyrus Mehta demonstrates how the 2009 article extends the Classical SWCI Method that was developed by Tsiatis, Rosner and Mehta in 1984; and then how he (along with Ping Gao and Lingyun Liu) extend this method to BWCI in order to include two-sided confidence intervals.
Related Items of Interest
[1] Brannath, Werner, Cyrus R. Mehta, and Martin Posch. "Exact confidence bounds following adaptive group sequential tests." Biometrics 65.2 (2009): 539-546. (Link directs to entire article, available via ResearchGate).
[2] Gao, Ping, Lingyun Liu, and Cyrus Mehta. "Exact inference for adaptive group sequential designs." Statistics in medicine 32.23 (2013): 3991-4005. (Link directs to entire article, available via ResearchGate).
