Adaptive Designs Are Re-Defining Drug Development – Learn What's New

Written by Jing Ping Yeo and Charles Warne
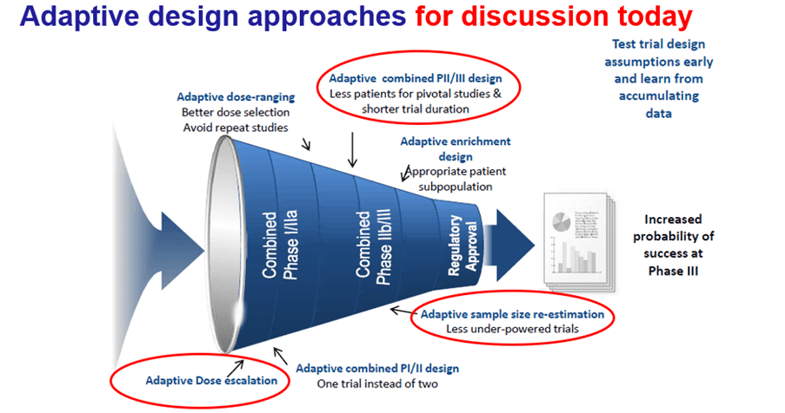
Adaptive designs are studies that “include a prospectively planned opportunity for modification of one or more specified aspects of the study design and hypotheses based on analysis of data (usually interim data) from subjects in the study,”1 with the aim to provide opportunity to gather more complete information about the therapeutic intervention from interim analyses. This lessens the unpredictability and reduces the risk of failure of Phase III confirmatory trials due to safety and efficacy concerns. In a traditional fixed-trial approach, the trial is designed and conducted as prescribed by the design until the end of the study when the final data are analyzed. Phases of clinical research are conducted as sequential, stand-alone studies from first-in-human Phase I dose escalation, to Phase II dose-finding, until final Phase III confirmatory trials leading to marketing approval. In adaptive trials, scheduled interim analyses are performed to learn from the accumulating data and make necessary modifications while the trial is ongoing, maintaining the study’s integrity and the validity of final conclusions. The most frequently appearing types of adaptations are seamless Phase II/III (57%), group sequential (21%), biomarker adaptive (20%), and adaptive dose-finding designs (16%).
In a recent DIA-Singapore conference presentation, Cytel biostatistician Charles Warne further elaborated on the three types:
1. Adaptive dose escalation
Mr. Warne reviewed the types of dose escalation methods, typically algorithm-based or model-based, with the most popular algorithm-based method being the 3+3 design. Such popularity is driven by its simplicity to understand and implement, yet the 3+3 method performs poorly on estimating the Maximum Tolerated Dose and limiting over/under dosing. These performance issues are not surprising given the 3+3 design uses fixed heuristic rules, rather than any statistical modelling to learn from the data already collected. This shortcoming is addressed with model-based dose escalation approaches. Cytel’s statisticians recently created the i3+3 method and helped popularize other methods, such as CRM and Bayesian Logistic Regression Models for combination studies. Middle ground ‘model-assisted’ approaches were discussed, such as mTPI and BOIN, which show similar performance to the model-based designs while being transparent for clinicians to understand and implement. The BOIN (which has been designated “Fit for Purpose” by the FDA) was compared with CRM, BLRM, and mTPI. Although BOIN is self-explanatory and user-friendly, trialists should always select the design best suited to their clinical context and research objectives.
2. Adaptive combined Phase II/III design
The inferentially seamless Phase II/III trial design was introduced. In this design, data from patients enrolled before (stage 1) and after (stage 2) adaption are combined for inferential confirmatory testing in the final analysis. A case study discussed some of the statistical issues to consider, such as controlling multiplicity due to multiple dose comparisons in stage 1 and combining data across stages in a statistically valid way that ensures strong Type I error control.
3. Adaptive sample size re-estimation
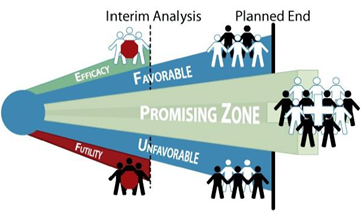
In the last segment of the presentation, the “promising zone design” for sample size re-estimation (SSR) was discussed. The SSR methods investigate the validity of the initial design assumptions based on accruing data and increase the sample size, if needed. This Promising Zone concept, developed by Cytel co-founder Dr. Mehta and colleagues, focuses on the interim analysis test statistic and its corresponding conditional power. Mr. Warne shared a case study, which demonstrated how the promising zone design is an attractive way for sponsors to gate their investment based on the accumulating data, so that the resources to increase sample size (and probability of success) are only committed when interim results are promising.
Overall, while there are important operational aspects to consider for adaptive designs, such designs can save time and cost by making what is an inherently complex drug development process more flexible to adapt to new information as it arises. Therefore, this design can be an innovative solution for drug developers in terms of resources and for patients in terms of health (i.e., getting improved and required treatment sooner and avoiding unnecessary exposure to the ineffective treatment).


Endnotes
1. U.S. Food and Drug Administration. (2010). Guidance for Industry: Adaptive Design Clinical Trials for Drugs and Biologics.

