
Single ascending dose (SAD) and multiple ascending dose (MAD) studies are typically the first in human studies. They seek to gain information on safety and tolerability, general pharmacokinetic (PK) and pharmacodynamic ( PD) characteristics, and of course identify the maximum tolerated dose (MTD).
Conventionally, SAD and MAD studies were conducted separately, but increasingly are combined into an ‘umbrella’ protocol which addresses both SAD and MAD objectives.
This approach can result in both time and cost savings, and allow additional valuable information to be gained earlier and inform subsequent development. When the studies are designed as a combined approach, adaptive principles are used as decisions will be made mid-study to adjust dose, change the number of subjects receiving a certain dose, or stop a treatment entirely.
In one recent project, we provided early phase trial design and biostatistics operational support for our client, an emerging biopharma with an early phase pipeline of products in neurological indications.
Challenge
With a constrained budget, and minimal infrastructure our client wanted an operationally efficient solution for a first-in-human study and day to day support from an expert team who would require minimal oversight.
Solution
Cytel statistical consultants devised an adaptive maximizing SAD/ MAD design combining multiple PK, PD, safety and tolerability objectives.
To create the study design, the Cytel consultant used Cytel’s specialist Compass software which is dedicated to the design, simulation, and execution of early phase clinical trials. It provides a systematic and efficient way to investigate and compare conventional versus adaptive dose-finding designs.
The below figure describes the structure of an example combined SAD/MAD approach.
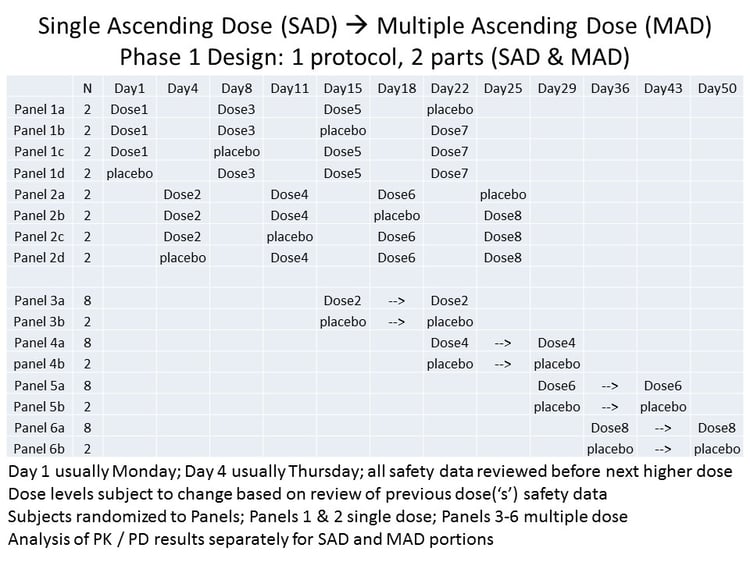
Cytel's experienced biostatistics and statistical programming team went on to provide analysis and reporting services for the project.
A SAD/MAD trial can be followed by an adaptive dose-finding trial to identify and solidify evidence of dose(s) with target level(s) of response.
Value added
The trial has been designed to provide a solid understanding of dose tolerability and PK/ PD measurements at doses with target levels of response. This enabled the client to move forward with a good regulatory package which provides the FDA and EMA the best evidence of early phase drug effect.
Further reading
A Competitive alternative to model based dose finding
Challenges in Neuroscience Clinical Trials
Adaptive dose-finding using toxicity probability intervals
Liked this? Download a .pdf copy of the case study to read offline.

Join our community of biopharma innovators and sign up for Cytel updates direct to your inbox.







