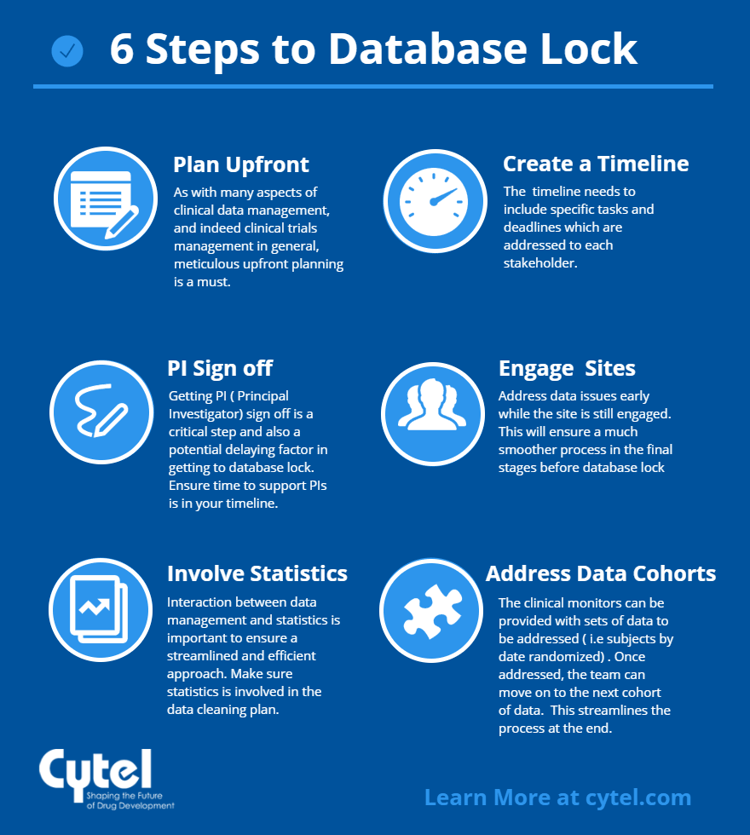
6 steps to timely database lock

To close a clinical database right the first time you have to begin with study start-up. Clearly, you can’t close a database if the data is not cleaned and you can’t have clean data unless you know what is most important for analysis. It’s imperative that data management works closely with the statistics group during CRF/ eCRF design to ensure data is being collected and data checks are being written in a meaningful fashion. But that’s still not enough. The data should be cleaned on a regular basis and forms locked as soon as the data has been SDVd and reviewed. Even then, it will be important to have your statistics team run listings and tables early on to catch anything unexpected. If the data is cleaned and locked by the time the last patient visit comes around then getting Principal Investigator sign-off and ultimately closing the database can run much more smoothly and quickly.
Cytel's ebook on optimizing data strategy provides an outline of the core tactics, tools, and tips for planning and executing a data strategy to boost the success of your clinical programs.
Database lock is a significant milestone in the clinical trial, upon which further data analysis and reporting timelines depend. The Clinical Data Manager is responsible for steering the data management process to ensure that the database is locked on time, and correctly. In this blog we lay out the 6 steps to database lock success.
Step 1: Planning
As with many aspects of clinical data management, and indeed clinical trials management in general, meticulous upfront planning is a must. As Stephen Covey famously put it:
"To begin with the end in mind means to start with a clear understanding of your destination. It means to know where you’re going so that you better understand where you are now so that the steps you take are always in the right direction."
There are a number of basics which must be observed and adhered to throughout the study as a matter of course:
- Creation of a solid database by ensuring proper data is being collected;
- Creation of solid data quality checks, ensuring they are written in a meaningful fashion;
- Creation of a comprehensive Data Management Plan, obtaining input from key stakeholders;
- Cleaning the data by addressing issues and queries on an ongoing basis; and
- Engaging on a regular basis with all stakeholders –clinical sites, statistics departments, sponsor and clinical operations
Step 2: Creating and managing a realistic timeline
A realistic timeline needs to be created to ensure the tasks involved are properly sequenced and concluded. With database lock relying on the input of a number of stakeholders, the timeline needs to include specific tasks and deadlines which are addressed to the specific groups involved.
At Cytel we would typically create a database close out check list which incorporates all of these items. Input and sign off to the check list and timeline should be obtained from clinical operations group and the sponsor.
At this point, it’s a matter of efficiently collecting and cleaning all the data which belongs in the Electronic data capture system and also conducting any reconciliation of data, such as laboratory data, which has been collected outside the EDC system.
Step 3: Obtaining timely sign-off from Principal Investigators (PIs)
Getting PI sign off is a critical step and also a potential delaying factor in getting to database lock. While PIs are trained in the appropriate EDC at the outset of the study, they tend not to be regularly working within the EDC system. This time lapse between the initial training and sign off / study close out, may mean that familiarity with the system is lost.
A useful way to handle this issue is to ensure that sufficient time to support the PIs around this activity is accounted for in the timeline.
Step 4: Engagement of Sites
One issue which can occur is when a clinical site might recruit all its patients at the beginning of the study. If the study continues for an extended time and the site has already completed their patients, the site may have little further interaction, and by the time of database lock they may have become somewhat disengaged from the study. In all, and especially in such, situations, it is advisable to address any data issues as early as possible while the site is still engaged. Keeping sites engaged as well as keeping up with data issues will make for a much smoother process in the final stages before database lock.
Step 5: Involving Statistics team
As we have explored in a previous blog, the interaction between Data Management and Statistics is an important one to ensure a streamlined and efficient approach. This is also true for all processes leading up to and including database lock. It’s important to ensure that the statistics group have been involved in the data cleaning plan as well as any data issues identified. By working jointly, the Clinical Data Management team can make sure the requirements of the statistics group are addressed.
Step 6: Tackling cohorts of data
One way in which database lock can be smoothed is to create cohorts of data – perhaps based on sets of subjects who were enrolled or randomized by a certain date. The clinical monitors can be provided with sets of data to be addressed, and once done, the team can move on to the next cohort of data. This is an efficient way to address large amounts of data and streamlines the process at the end.
In summary, the key areas which will ensure a smooth conclusion to the data collection process are planning and engagement. With strong attention to detail, timelines, and ongoing involvement with the key stakeholders we can ensure the database is locked on time, or even ahead of schedule.
See our infographic below:
With thanks to Patti Arsenault, Director Clinical Data Management at Cytel.
Read more from Perspectives on Enquiry and Evidence
Sorry no results please clear the filters and try again

Can RWE Help Restore Decades of Health Inequalities? Yes, and Here’s How

FDA Guidance on the Design and Conduct of Externally Controlled Trials — What to Watch



