5 Reasons to Invest in Adaptive Designs for Population Enrichment
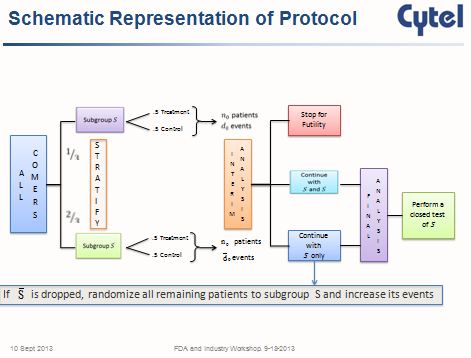
The above graphic is from Cyrus Mehta's slides on 'Adaptive Population Enrichment for Oncology Trials with Time to Event Endpoints.'
Recent advances in precision medicine have meant that therapeutic treatments can now target subsets of a population that are most likely to respond well to treatment. Identification of such subsets largely relies on the presence or absence of particular biomarkers. In order to determine whether or not such biomarkers have predictive diagnostic capabilities, the biomarkers must first be validated as reliable predictive indicators, and thereafter as responding efficaciously to treatment.
An adaptive enrichment design creates a single trial in which a study team can accomplish the combined task of validating a predictive biomarker and evaluating for efficacy. Flexibility of design choices allows evaluations of efficacy to take place simultaneously for both the full population and the subpopulation with the presence of the biomarker. While there are many types of adaptive enrichment designs, one popular amongst Cytel’s clientele begins with a sequential trial with the full population segmented by biomarker status. Interim analyses then determine whether the therapy appears favorable or promising for the full population or in the biomarker-defined subpopulation.At this point several states of affairs might arise. For example, results could show that:
Given these various interim outputs, trial designers might consider whether it is worthwhile to invest in adaptive enrichment designs. Here are five reasons why such designs may be worthwhile for your next trial:
Uncertainty about the Effects of Prognostic Biomarkers:
In a recent interview in Nature Review Drug Discovery, Tatiana Prowell of the FDA cited the following concern. ‘A trial may find that patients with a certain biomarker get a drug and have a good outcome, but this might be because patients with this biomarker do better than patients without it in general.’ [1] If a trial enters Phase 3 with uncertainty about the general benefits to a patient of possessing a prognostic biomarker, then validating the same biomarker’s predictive capabilities becomes problematic (i.e. it is unclear whether the positive results are due to the effects of predictive enrichment, or the general prognostic benefits of having the biomarker in question.) An adaptive design makes it possible to compare a control group with the biomarker subpopulation to a control group for the full population, ensuring that prognostic biomarkers that are also predictive do not confuse study outcomes.
Financial Risk-Mitigation through Enrichment:
When entering a Phase 3 trial with underwhelming results from Phase 2, it may be worthwhile to aim to mitigate the risk of Phase 3 failure. An adaptive enrichment design offers one strategy of doing so. Predictive enrichment can rescue a trial in situations where unfavorable results are determined for the full-population, if promising or favorable results are determined for the biomarker subpopulation. In such cases, an adaptive design offers an option to continue the study using only the trial arms with a biomarker subpopulation.
Incentivizing Investors:
There are obvious difficulties with finding investors for large Phase 3 trials, particularly in therapeutic areas where trials often fail. Favorable interim results for biomarker subpopulations signal to investors that the drug in question will likely make it to market regardless of the trial outcome for the full-population. This incentivizes investment in the trial.
[In fact, adaptive designs have been successfully used by Cytel’s clients to mitigate financial risk, by allowing for investments that are conditional upon interim results. For more information about such procedures, view this video of the VALOR trial.]
Protecting Patients:
When discussing predictive enrichment, the discussion often pivots around identifying subpopulations with predictive biomarkers whose response to therapy is better than that of the full population. However, from the patient perspective, it is equally important to know if he or she possesses a biomarker that is less likely to respond to a therapeutic treatment that is offered to the full population. Collecting such data in non-adaptive trials would require waiting for trial completion before conducting analysis. This would likely take several years, during which time other people with the biomarker might be enrolled on the same trial. An adaptive design, by contrast, can study the biomarker subpopulation in question, establish that the subpopulation should not be taking the drug, and then proceed with the remaining clinical arms.
Data Collection as a Secondary Objective for Future Trials:
An adaptive enrichment design allows for a sequential trial where a full population is segmented during interim analyses. As a result, data about the effects of a drug on a particular biomarker may already be available (or at least readily accessible) while completing a trial on the full-population. If there are many biomarkers of interest, then an adaptive trial (combined with appropriate pre-specification) can play the dual role of determining efficacy for the full-population while gathering data on subpopulations. The data-gathering might be a secondary objective, but can provide useful information for future trials. For example, while retrospective discoveries of correlation might raise concerns about the spuriousness of the correlation in question, an adaptive trial will ensure that the biomarker subpopulation is clearly compared to the appropriate control group.
*Many thanks to Cytel Consulting Senior Director Zoran Antonijevic for comments on this post.
Related Items of Interest
Cyrus Mehta, et al., 'Biomarker Driven Adaptive Population Enrichment for Oncology Trials with Time to Event Endpoints,' (forthcoming: Statistics in Medicine) Paper, Slides
Adaptive Designs for Precision Medicine: A Look at Pfizer's Xalkori Trial
Overcoming Prowell's Pitfalls: Cytel Weighs in on Strategies for Oncology Drug Development
Powering Oncology Trials for Success
Cytel Joins DIA Discussion on Predictive Enrichment
5 Reasons to Invest in Bayesian Dose-Escalation


