Impact of Study Design and Development Strategy on Pharmaceutical Programs and Portfolios
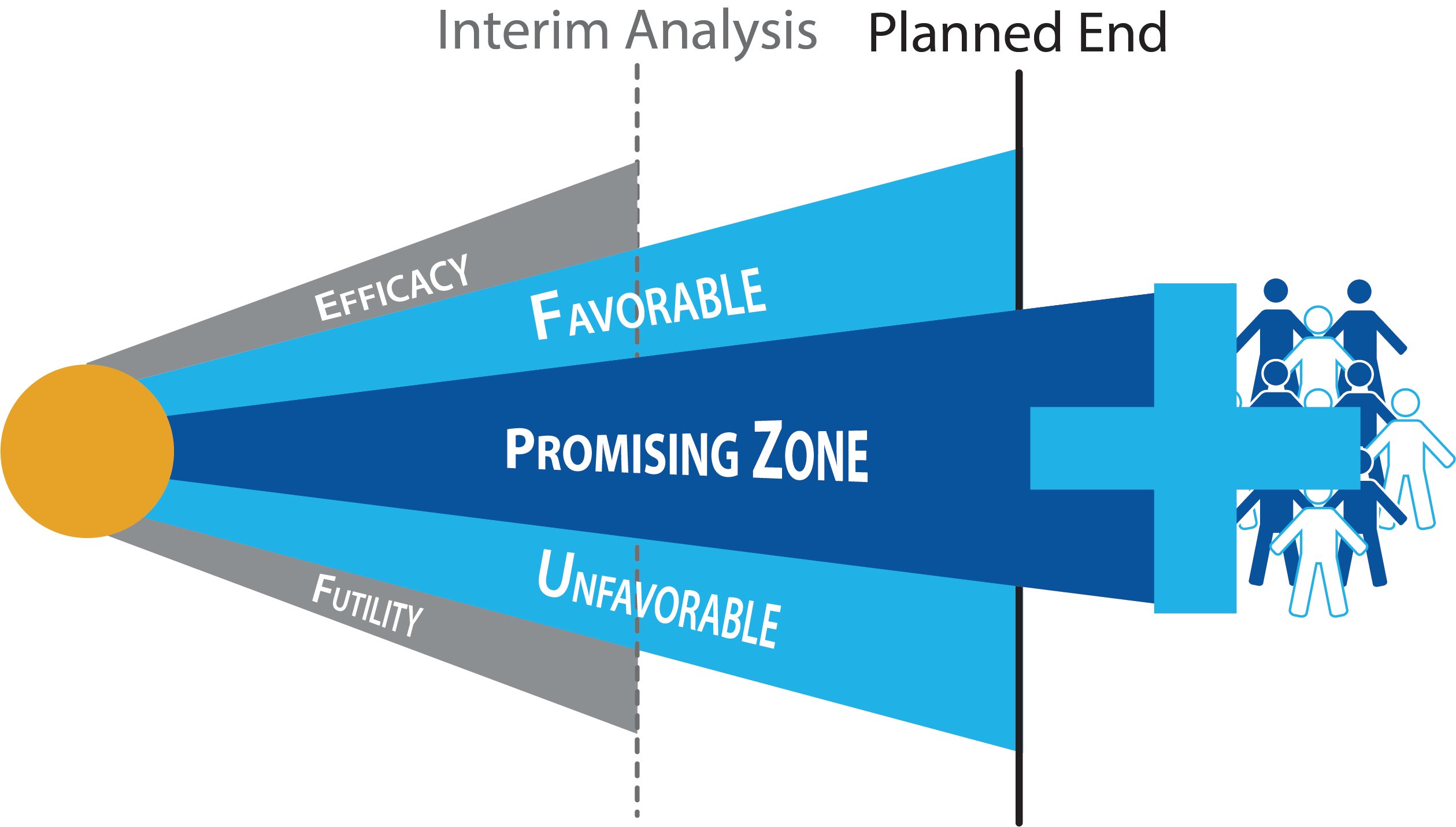
As more clinical trials make use of adaptive designs, investors have come to realize that high quality trial designs can result in significant improvements to a trial’s financial risk profile. Regardless of a trial’s eventual success or failure, a well-constructed design provides a drug with the highest possible probability of success while mitigating financial risk.
Consider Sunesis’s VALOR trial which will be unblinding later this year. According to Sunesis CMO Adam Craig, the adaptive promising zone design proved instrumental for acquiring investment from Royalty Pharma. Royalty Pharma agreed to provide Sunesis $25 million in return for 3.6% of net sales conditional upon the trial continuing after the interim look. If the interim look resulted in a sample size re-estimation, then Royalty Pharma’s returns would increase to 6.75% of net sales to account for increase in risk.
An extension of this concept provides a general framework for determining the impact of study design and development strategy on pharmaceutical programs and portfolios. In a soon to be released volume entitled, ‘Optimization of Pharmaceutical R&D Programs and Portfolios: Design and Investment Strategy,’ an array of experts from the pharmaceutical industry demonstrate the role of study design in optimizing the value of a program or a portfolio. You can find details of the new book below. It will be available from Springer in mid-September.
Clinical Development Strategy
Optimization of Pharmaceutical R&D Programs and Portfolios: Design and Investment Strategy
Edited by Zoran Antonijevic, Senior Director at Cytel Consulting
Summary
Very little has been published on optimization of pharmaceutical portfolios. Moreover, most of the published literature comes from the commercial perspective, where probability of technical success (PoS) is treated as fixed, and not as a consequence of development strategy or design. In this book there is a strong focus on the impact of study design on PoS, and ultimately a portfolio’s value. Design options that are discussed are dose-selection strategies, adaptive design, and enrichment. Some development strategies that are examined are indication sequencing, optimal number of programs, and optimal decision criteria.
This book includes chapters written by authors with very broad backgrounds including financial, clinical, statistical, decision sciences, commercial, and regulatory. Many authors have long held executive positions and have been involved with decision making at a product or at a portfolio level. As such, it is expected that this book will attract a very broad audience, including decision makers in pharmaceutical R&D, commercial, and financial departments. The intended audience also includes portfolio planners and managers, statisticians, decision scientists, and clinicians.
Early chapters describe approaches to portfolio optimization from big pharma, and venture capital standpoints, focusing on finances and processes. Later chapters present selected statistical and decision analysis methods for optimizing drug development programs and portfolios. Some methodological chapters are technical; however, with a few exceptions they require a basic knowledge of statistics by a reader.
Introduction
1. Zoran Antonijevic, Cytel
Need for Optimal Design of Pharmaceutical Programs and Portfolios in Modern Medical Product Development.
Background
2. Rick Sax, Quintiles
Clinical Aspects of Pharmaceutical Portfolio Management
3. Kraig Schulz, Ernst & Young
Drug Development and the Cost of Capital
4. Ray Huml, Quintiles
Investment Considerations for Pharmaceutical Product Portfolios
5. Charles Persinger, Lilly
Challenges of Portfolio Management in Pharmaceutical Development;
Quantitative Methodology
6. Zoran Antonijevic, Cytel
Impact of Phase 2b Strategies on Optimization of Drug Development Programs
7. Richard Nixon, Novartis
Using Decision Analysis to Support the Design of Clinical Trials at a Program Level;
8. Jack Kloeber, Kromite,
Indication Sequencing for a New Molecular Entity with Multiple Potential Oncology Indications
9. Cong Chen, Merck,
Maximizing Return on Investment in Phase II Proof-of-Concept Trials
10. Bob Beckman, Daiichi-Sankyo, Inc.
Portfolio Optimization of Therapies and Their Predictive Biomarkers
11. Nitin Patel, Cytel,
Dynamically Optimizing Budget Allocation for Phase 3 Drug Development Portfolios Incorporating Uncertainty in the Pipeline

